TL;DR:
- The 2026 regulatory overhaul introduces stricter, structurally different standards for consumer goods in the EU and US.
- Key requirements include safety assessments, sustainability data, and PFAS substance bans across multiple sectors.
- Proactive compliance, integrated into product design from the start, provides a competitive advantage and reduces risks.
Even well-established brands can find themselves locked out of major markets overnight when compliance standards shift. The 2026 regulatory cycle is the most disruptive in over a decade, bringing sweeping changes to how consumer goods are formulated, documented, labeled, and sold across the EU and US. From PFAS chemical bans to Digital Product Passports, the rules are not just stricter. They are structurally different. This guide breaks down what changed, why it matters by sector, and exactly what your team needs to do to stay market-ready and audit-proof in 2026.
Table of Contents
- The 2026 compliance landscape: What's changed and why it matters
- Core requirements: Safety, sustainability, and PFAS restrictions
- Key 2026 requirements by sector: Cosmetics, electronics, textiles, and more
- Documentation, labeling, and cost: Your compliance execution checklist
- Our take: Compliance success means creative adaptation, not box-ticking
- How FormlyPro empowers your 2026 compliance journey
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| 2026 mandates overhaul | New safety, chemical, and sustainability standards mean every brand must re-evaluate compliance processes. |
| Sector rules vary | Electronics, cosmetics, and textiles each face unique 2026 compliance requirements and bans. |
| Documentation is critical | Failing to provide traceable documentation and labels can instantly block market access. |
| Compliance costs add up | Testing and certification for market entry can range from $5,000 to $25,000 per model. |
| Adaptation is key | Integrating compliance early in design gives brands a competitive edge under 2026 rules. |
The 2026 compliance landscape: What's changed and why it matters
The regulatory environment for consumer goods in 2026 is not a gradual evolution. It is a reset. Two frameworks are reshaping how products enter and stay in European markets. The EU GPSR and Digital Product Passport (DPP) now govern nearly all non-food consumer goods sold in the EU, replacing older, patchwork directives with unified, enforceable standards. These are not optional guidelines. They carry real enforcement teeth, including import blocks, mandatory recalls, and fines.
On the chemical side, PFAS restrictions are expanding rapidly in both the US and EU, with significant effective dates in 2026 and 2027. These synthetic chemicals, once standard in everything from cookware coatings to water-resistant textiles, are now being phased out under regulatory pressure that shows no signs of slowing down.
The EU approach leans toward harmonization. One set of rules, applied consistently across all member states. The US, by contrast, is a patchwork. California, New York, and Maine are each enforcing different PFAS timelines. Washington state has its own requirements for children's products. Navigating fragmentation vs. harmonization is itself a full-time compliance challenge for brands selling across North America.
The consequences of falling short are significant:
- Market exclusion: Products that miss DPP or GPSR requirements cannot legally be sold in the EU.
- Recall costs: Product recalls triggered by documentation gaps or undisclosed substances can run into the millions.
- Reformulation penalties: Brands discovered using restricted substances face mandatory reformulation timelines that delay launches.
- Reputational damage: Public recall notices and enforcement actions are searchable forever.
More brands are now discovering that compliance is not just a legal function. It is a launch function. Understanding the full scope of 2026 consumer goods standards before you finalize a formulation is what separates brands that scale from brands that stall.
With the context set for what's at stake, let's break down the major categories of standards no product brand can ignore.
Core requirements: Safety, sustainability, and PFAS restrictions
Think of 2026 compliance as three intersecting pillars: safety, sustainability, and chemical restrictions. Each one has its own documentation requirements, enforcement timeline, and cost implications.

Safety under the GPSR mandates that every non-food product sold in the EU comes with a formal risk assessment, technical documentation, and a clear traceability chain. That means batch-level records, market surveillance obligations, and a named responsible economic operator in the EU if you are a non-EU brand. The days of submitting a generic safety declaration and calling it done are over.

Sustainability is now a product-level data requirement. The DPP requires machine-readable, traceable lifecycle and environmental data attached to each product in key sectors starting in 2026. This is not marketing copy. It is structured data accessible via QR code or RFID that regulators and consumers can verify. If your supply chain cannot produce this data, you cannot produce a compliant product.
| Compliance pillar | Key requirement | Documentation format |
|---|---|---|
| Safety (GPSR) | Risk assessment, technical file | PDF technical dossier, EU declaration |
| Sustainability (DPP) | Lifecycle data, environmental metrics | Machine-readable data carrier |
| PFAS restrictions | Substance-free certification | Lab test reports, supplier declarations |
PFAS bans are the third pillar. PFAS expansion in 2026 covers textiles, electronics, cosmetics, and select food contact materials across both the US and EU. France has already enacted standalone PFAS restrictions in cosmetics. The EU's universal restriction proposal covers thousands of PFAS substances. The US is moving state by state but converging on similar outcomes.
"Brands that treat PFAS compliance as a single regulatory checkbox will be reformulating again in 18 months. The phase-out is structural, not episodic."
Pro Tip: Start your PFAS audit at the raw material level, not the finished product. Many ingredient suppliers have not updated their safety data sheets to reflect 2026 restriction lists. Cross-reference your input materials against both ECHA candidate lists and current US state banned substance lists before finalizing any formula.
You can map your specific compliance obligations using this compliance checklist and understand how to integrate these requirements in your workflow through this guide on compliance in product development.
Understanding the broad requirements, the next layer is how these standards play out for specific product categories where rules are most stringent.
Key 2026 requirements by sector: Cosmetics, electronics, textiles, and more
Compliance is not one-size-fits-all. The rules that apply to a facial serum are different from those governing a smart wearable or a performance jacket. Here is what each major category faces in 2026.
Cosmetics are under significant pressure from Omnibus VIII (EU 2026/78), which applies from May 2026 and restricts a new wave of CMR (carcinogenic, mutagenic, and reprotoxic) substances, introduces tightened nano-particle rules, and expands traceability requirements for responsible persons. If your formulation includes fragrance allergens, certain preservatives, or UV filters under review, re-evaluation is urgent.
Electronics are the first sector to face mandatory DPP digital documentation, with sustainability metrics including repairability scores, recycled content percentages, and carbon footprint data now required. Labeling requirements are also expanding to include energy class updates and end-of-life instructions.
Textiles must comply with PFAS bans in multiple jurisdictions while simultaneously preparing for digital passport rollout. Water and stain resistance claims built on PFAS chemistry must be reformulated or dropped.
| Sector | Key 2026 requirement | Effective date |
|---|---|---|
| Cosmetics | CMR substance bans, nano rules (Omnibus VIII) | May 2026 |
| Electronics | DPP data carrier, sustainability metrics | 2026 phase-in |
| Textiles | PFAS ban, digital passport prep | 2026-2027 |
| Food packaging | PFAS trace limits, substance risk review | 2026+ |
Here are the steps most sector teams are using to prioritize their 2026 compliance work:
- Audit current formulations or materials against updated restricted substance lists.
- Identify which DPP sectors your product falls into and map the data requirements.
- Commission third-party lab testing for PFAS presence in finished products and key inputs.
- Update supplier agreements to include substance restriction warranties.
- Build internal documentation workflows that can produce audit-ready files on demand.
For deeper detail on cosmetics-specific rules, this resource on cosmetics compliance covers the Omnibus VIII changes in practical detail. You can also review formulation compliance case studies to see how other brands have navigated similar restrictions.
With sector specifics clarified, brands must understand universal documentation, labeling, and cost realities close up.
Documentation, labeling, and cost: Your compliance execution checklist
Compliance paperwork is not bureaucratic overhead. It is your defense against recalls, import blocks, and enforcement actions. Every product in 2026 needs a traceable identity: batch codes, QR-linked data, ingredient transparency, and appropriate regulatory marks like CE for EU markets.
FDA labeling requirements and equivalent EU standards mandate that product labels include complete ingredient lists, country of origin, responsible party contact, and any required warnings. For products crossing multiple jurisdictions, this means managing two or more label versions with legal review for each.
On cost, budget realistically. Compliance testing for regulated consumer products typically runs between $5,000 and $25,000 per model, depending on the category and number of standards involved. Electrical safety, chemical substance testing, and DPP data infrastructure each carry separate costs.
The 5-stage compliance process that leading brands follow looks like this:
- Identify standards: Map every applicable regulation by market and category before development begins.
- Assess risk: Run formal risk assessments against current restricted substance lists.
- Test and certify: Commission accredited lab testing and obtain required certifications.
- Document: Build a technical file that covers design, materials, test results, and post-market monitoring plans.
- Monitor: Establish a process for tracking regulatory updates and triggering re-review when rules change.
Pro Tip: Do not wait for a product launch to discover a documentation gap. Run a mock audit six weeks before any planned launch date. Have someone outside your team attempt to reconstruct your compliance file from scratch. Gaps become obvious fast.
Without clear traceability, recall liability extends across your entire supply chain. The UN principles for consumer product safety emphasize that business lifecycle responsibility does not end at the point of sale. Tracking matters at every stage.
You can find a detailed breakdown of the process in this guide on market-ready formulation steps.
With a practical checklist in hand, let's address what many guides miss: the real-world friction and how leading brands actually achieve compliance.
Our take: Compliance success means creative adaptation, not box-ticking
Most brands approach compliance as a gate to pass through before a product can launch. That framing creates blind spots. It pushes compliance to the end of the development cycle, where it becomes a bottleneck and a cost center rather than a strategic input.
The brands that consistently win at market entry treat compliance as a design constraint, not a finish-line hurdle. When regulatory requirements inform ingredient selection, packaging design, and data architecture from day one, the audit-ready file almost builds itself. The teams that adapt this way move faster, not slower.
What the 2026 rules demand is digital traceability at scale. That is a capability, not a checkbox. Brands that build that capability now, across formulation, labeling, and supply chain documentation, will have a structural advantage over competitors who are still reformulating reactively in 2027.
Understanding why compliance matters is not just about avoiding fines. It is about building a product infrastructure that can withstand market scrutiny and scale across jurisdictions without starting over each time.
Now that you've gained both frameworks and honest perspective, see how specialized support can streamline your 2026 compliance journey.
How FormlyPro empowers your 2026 compliance journey
Staying on top of shifting regulations while simultaneously developing new products is a real operational challenge. FormlyPro is built specifically for product developers and brand owners who need both strategic clarity and execution support in one place.
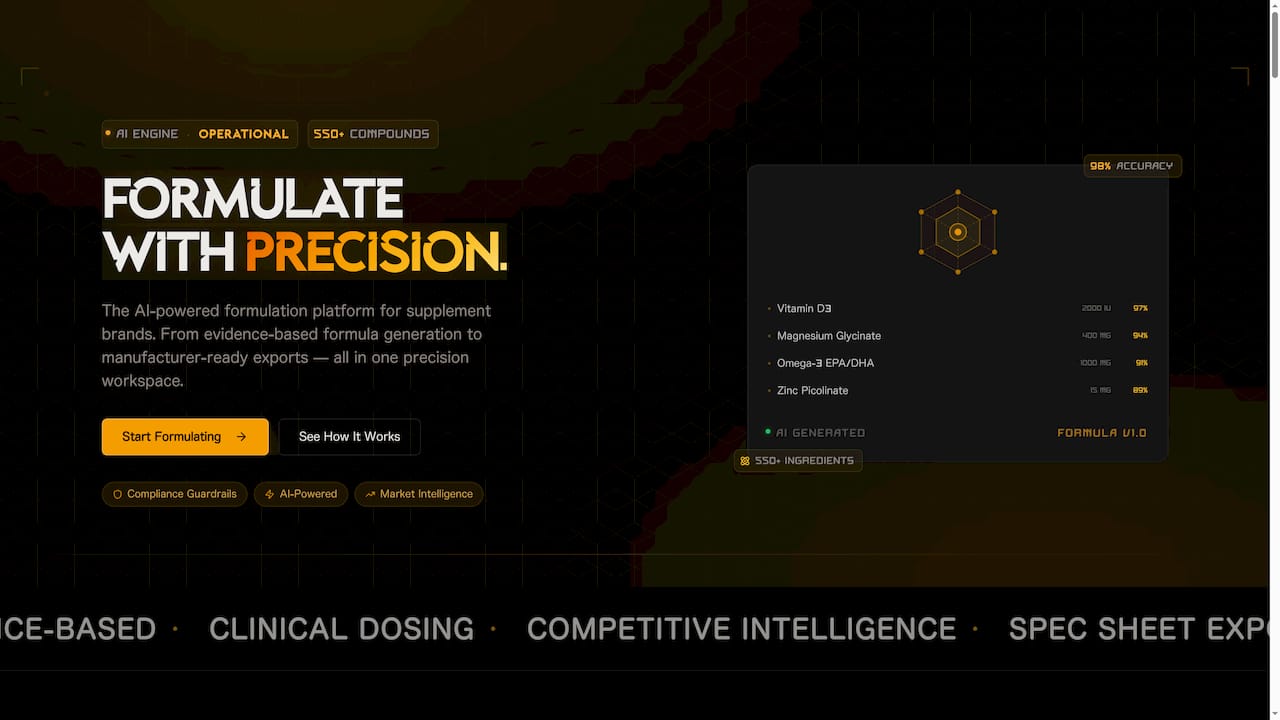
The platform automates formula compliance checks against current restricted substance lists, tracks regulatory updates across the EU and US, and generates the traceability documentation your team needs to pass audits. Its 8-phase product development plan walks you from initial ideation through formulation, compliance review, and production, with built-in market research and competitor analysis at every stage. If you are building or reformulating a product for 2026 market entry, this brand formulation guide shows exactly how the platform supports your compliance workflow from the ground up.
Frequently asked questions
What are the new 2026 PFAS restrictions for consumer goods?
PFAS bans take effect in US states and across the EU for textiles, cosmetics, and electronics starting January 2026, with broader EU-wide restrictions scheduled to follow. Brands should audit current formulations against both regional and sector-specific banned substance lists immediately.
Who must comply with EU Digital Product Passport rules in 2026?
Manufacturers and brand owners in electronics, textiles, and toys must begin DPP implementation in 2026, with sector-specific data requirements phasing in between 2026 and 2027. Non-EU brands selling into the EU market are equally obligated.
How much does product compliance testing cost in 2026?
Compliance testing costs typically range from $5,000 to $25,000 per model for consumer electronics and other regulated product categories. Budget early, as testing timelines can add four to eight weeks to a launch schedule.
What documentation is mandatory under the 2026 EU GPSR?
Under GPSR requirements, manufacturers must provide a formal technical risk file, transparent labeling, and a clear traceability chain for all non-food consumer products sold in EU markets. A named EU-based responsible economic operator is also required for non-EU brands.
