Compliance is one of those words that makes founders tense up. Many entrepreneurs treat it as a legal checkbox, something to deal with after the product is already built. That instinct is costly. The brands that scale fastest in the food and supplement space are the ones that bake compliance into their strategy from day one, not because they fear regulators, but because they understand that a clean regulatory record is a genuine business asset. This guide breaks down what compliance actually means, why it drives growth, and where the hidden traps are that catch even experienced brands off guard.
Table of Contents
- What compliance really means in food and supplements
- Critical reasons why compliance is important
- Hidden risks and edge cases: Social media, CBD, imports
- Modernization and the future of compliance
- A smarter approach: Make compliance your competitive edge
- Streamline your compliance journey with FormlyPro
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Compliance builds trust | Meeting regulations assures customers and partners your products are safe and credible. |
| Non-compliance is costly | Ignoring rules exposes brands to recalls, legal action, and lost sales opportunities. |
| Stay ahead of change | Adapting early to new compliance rules makes your business resilient and ready for growth. |
| Turn compliance into an advantage | Promote your compliance efforts to stand out and gain access to new markets and retailers. |
What compliance really means in food and supplements
Compliance is not just about avoiding trouble. At its core, it means your product, your label, and your marketing all meet the legal standards set by the agencies that oversee your category. In the United States, that primarily means the Food and Drug Administration (FDA) and the Federal Trade Commission (FTC). The FDA focuses on product safety, labeling accuracy, and manufacturing standards. The FTC governs advertising claims, especially when those claims could mislead consumers.
For supplement brands specifically, the key law is the Dietary Supplement Health and Education Act, known as DSHEA. Unlike pharmaceutical drugs, DSHEA governs supplements through post-market enforcement rather than pre-market approval. That means you do not need FDA sign-off before launching, but you are fully responsible for the safety and accuracy of everything you put on the market. The FDA can act after the fact if problems arise.
Food brands face a slightly different framework. Certain ingredients require pre-market approval as food additives, while generally recognized as safe (GRAS) ingredients have a different pathway. New dietary ingredients (NDIs) in supplements require a notification to the FDA before use.
Here is a quick breakdown of the main compliance categories every brand must understand:
| Compliance area | What it covers | Key agency |
|---|---|---|
| Safety | Ingredient safety, NDI notifications, GRAS status | FDA |
| Labeling | Nutrition facts, structure/function claims, disclaimers | FDA |
| Marketing claims | Advertising truthfulness, substantiation | FTC |
| Manufacturing | Good Manufacturing Practices (GMPs) | FDA |
| Import/Export | Country of origin, ingredient documentation | FDA, CBP |
The most common compliance requirements brands encounter include:
- Accurate nutrition and supplement facts panels
- Proper structure/function claim disclaimers (the "This statement has not been evaluated" language)
- GMP-certified manufacturing facilities
- NDI notifications for novel ingredients
- Substantiated advertising claims backed by evidence
Understanding these categories upfront saves you from expensive reformulations and label reprints later.
Critical reasons why compliance is important
Compliance is not just a legal shield. It is a growth lever. The supplement industry has grown into a $60B+ market largely because DSHEA created a flexible regulatory environment that allowed innovation while maintaining consumer protections. That market exists because of the regulatory framework, not in spite of it.

Here is a direct comparison of what compliance delivers versus what ignoring it costs:
| With compliance | Without compliance |
|---|---|
| Retailer and distributor access | Rejected by major retail buyers |
| Consumer trust and repeat purchases | Damaged brand reputation |
| Legal defensibility | Exposure to lawsuits and FDA action |
| Smooth import/export operations | Shipment holds and import bans |
| Investor and partner confidence | Difficulty raising capital |
Retailers like Whole Foods, Target, and Amazon all have compliance requirements before they will stock a product. A single warning letter from the FDA can disqualify you from those shelves. That is not a theoretical risk. It happens regularly.
The consequences of ignoring compliance stack up fast:
- Product recalls that cost tens of thousands of dollars and destroy consumer confidence
- FDA warning letters that become public record and follow your brand permanently
- FTC enforcement actions with financial penalties for misleading advertising
- Import detentions that freeze your supply chain
- Loss of retail partnerships that took years to build
Pro Tip: Do not treat compliance as a phase you complete before launch. Build it into your product development timeline from the first formulation meeting. It is far cheaper to get it right at the start than to fix it after you have already printed 10,000 labels.
The brands that win long-term in this space understand that compliance is a form of quality signaling. It tells retailers, consumers, and investors that you operate with integrity.

Hidden risks and edge cases: Social media, CBD, imports
Most brands understand the basics of labeling and manufacturing compliance. What trips them up are the edge cases, the compliance risks hiding in places they never thought to look.
Social media is one of the biggest blind spots. Social media claims, curated reviews, and CBD content are among the most common triggers for FDA warning letters in 2025 and 2026. If an influencer you paid posts a claim that your product "cures" or "treats" a condition, that post is legally your responsibility. The FDA does not distinguish between your website copy and a sponsored Instagram story.
"Warning letters increasingly cite third-party content, including influencer posts and customer review pages, as evidence of illegal disease claims. Brands are caught off guard because they assume only their own website copy is under scrutiny."
Here are four hidden compliance traps that catch brands by surprise:
- Social media health claims: Any post, story, or video that implies your product treats a disease can trigger enforcement, even if a third party made the claim on your behalf.
- CBD and delta-8 products: These remain in a legal gray zone at the federal level. Marketing them as dietary supplements is not currently permitted under FDA guidance.
- Import and ingredient documentation: If your raw materials come from overseas suppliers, you are responsible for verifying their safety and documentation. A supplier's certificate of analysis is not enough on its own.
- White-label and private label liability: If you put your brand name on a product, you own the compliance responsibility, even if you did not formulate it.
Pro Tip: Schedule a quarterly review of all third-party content associated with your brand, including influencer posts, affiliate sites, and retailer product pages. What you do not catch, the FDA might.
Modernization and the future of compliance
The regulatory landscape for supplements is not static. There are active debates in Washington about how to modernize DSHEA to address the sheer volume of products now on the market. The current modernization debates center on mandatory product listing requirements, not full pre-market approval. That is an important distinction. Mandatory listing would require brands to register their products with the FDA, giving regulators better visibility without creating a drug-style approval bottleneck.
The scale of the challenge is real. Thousands of new supplement products enter the U.S. market every year, and the FDA's post-market enforcement resources are stretched thin. Brands that voluntarily maintain strong documentation and transparent practices are far less likely to face scrutiny.
Here are three steps you should take now to stay ahead of where compliance is heading:
- Track regulatory guidance actively. Subscribe to FDA and FTC updates. Follow industry organizations like the Council for Responsible Nutrition (CRN) to stay informed on proposed rule changes before they become law.
- Implement lot-level documentation. Maintain batch records, certificates of analysis, and supplier documentation for every production run. This is already required under GMPs, but many small brands are not doing it consistently.
- Engage qualified regulatory counsel early. A regulatory attorney or consultant who specializes in food and supplements is an investment that pays for itself the first time they catch a label issue before it goes to print.
Brands that build these habits now will have a significant advantage when new requirements take effect. Reactive compliance is always more expensive than proactive compliance.
A smarter approach: Make compliance your competitive edge
Here is the uncomfortable truth most compliance conversations skip: the brands that treat compliance as a burden are the ones that get burned. The brands that treat it as a strategic investment are the ones that end up on Whole Foods shelves, in major retail chains, and in front of serious investors.
We have seen this play out repeatedly. A startup with a genuinely innovative formulation gets rejected by a major distributor because their label has an unsubstantiated claim. Meanwhile, a less innovative competitor with airtight compliance documentation walks straight through that same door. The product quality was not the deciding factor. The paperwork was.
That is a hard lesson, but it points to a real opportunity. If you can demonstrate clean compliance records, third-party testing, and documented manufacturing standards, you have a pitch that most of your competitors cannot match. Retailers and distributors are increasingly asking for this documentation upfront. Being able to hand it over immediately signals that you are a serious, scalable brand.
Pro Tip: Include a one-page compliance summary in your retailer pitch deck. List your GMP certifications, third-party testing partners, and any NDI notifications filed. It shows buyers you have done the work and reduces their due diligence burden significantly.
Compliance is not the ceiling on your ambition. It is the foundation that lets you build higher.
Streamline your compliance journey with FormlyPro
If navigating all of this feels overwhelming, you are not alone. Most founders are experts in their product, not in regulatory frameworks. That is exactly the gap FormlyPro is built to close.
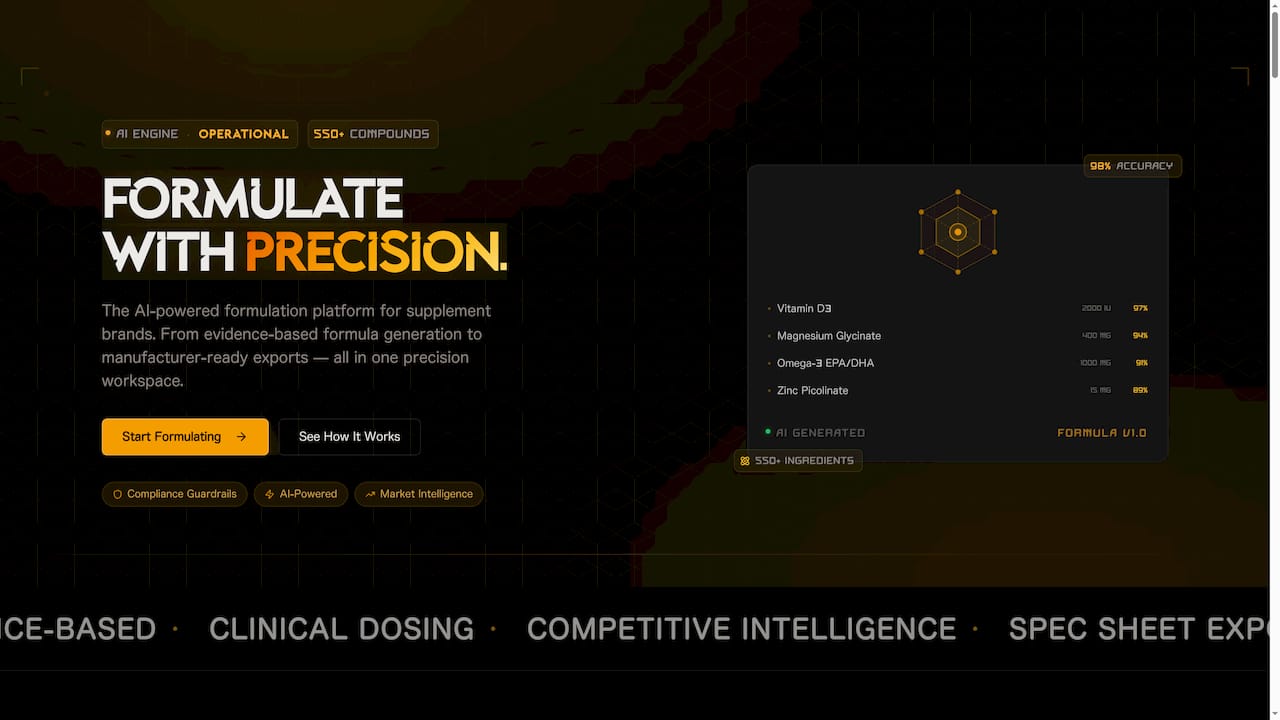
FormlyPro guides food and supplement brands through every compliance requirement tied to their specific product and category, from pre-market planning and label review to ongoing monitoring and export readiness. The platform is built into an 8-phase product development system that takes you from initial concept through formulation, compliance, and production, with market research and competitor analysis built in at every step. You also get access to an AI-powered packaging mockup designer so your brand looks as good as it performs on shelf. If compliance has been the thing slowing you down, FormlyPro is where you start.
Frequently asked questions
What is DSHEA and how does it affect my supplement business?
DSHEA provides the legal framework for the U.S. supplement market, relying on post-market enforcement rather than pre-approval, which gives brands flexibility while requiring truthful labeling and safe ingredients. Understanding it is non-negotiable for any supplement brand operating in the U.S.
Can making claims on social media put my brand at risk?
Yes. Social media claims are on FDA's radar, and brands have received warning letters for influencer posts and curated customer reviews that made unsupported health claims. You are responsible for third-party content you facilitate or pay for.
What are the consequences of non-compliance?
Failure to comply can result in product recalls, import bans, FDA warning letters, FTC penalties, and lasting reputational damage that cuts off retailer and investor relationships.
How can brands prepare for future compliance changes?
Monitor calls to modernize DSHEA such as mandatory product listing proposals, keep your documentation practices current, and work with a regulatory specialist who tracks these developments closely.
