70% of new food products fail within their first two years on shelf. That number is even grimmer when you factor in beauty and wellness launches, where consumer expectations shift fast and regulatory scrutiny is tightening. The brands that survive aren't just lucky. They follow a structured, data-driven process that connects market insight to formulation decisions, compliance to packaging, and prototyping to post-launch monitoring. This guide walks you through every critical phase, so you can move from a raw concept to a compliant, market-ready product without burning your budget on avoidable mistakes.
Table of Contents
- Step 1: Market analysis and idea validation
- Step 2: Formulation design and prototyping
- Step 3: Safety, sensory, and regulatory testing
- Step 4: Scale-up, packaging, and launch monitoring
- Our perspective: Why a data-driven approach beats guesswork
- Streamline your formulation journey with FormlyPro
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Market analysis is critical | Deep research and competitive benchmarking drastically improve your formula's market fit. |
| Data-driven prototyping saves time | Using AI and analytics cuts prototypes and costs, reducing timelines by 50% or more. |
| Compliance can’t be skipped | Documenting safety and regulatory steps from the start prevents costly recalls and legal issues. |
| Continuous improvement wins | Ongoing monitoring and adaptation after launch are key to long-term brand success. |
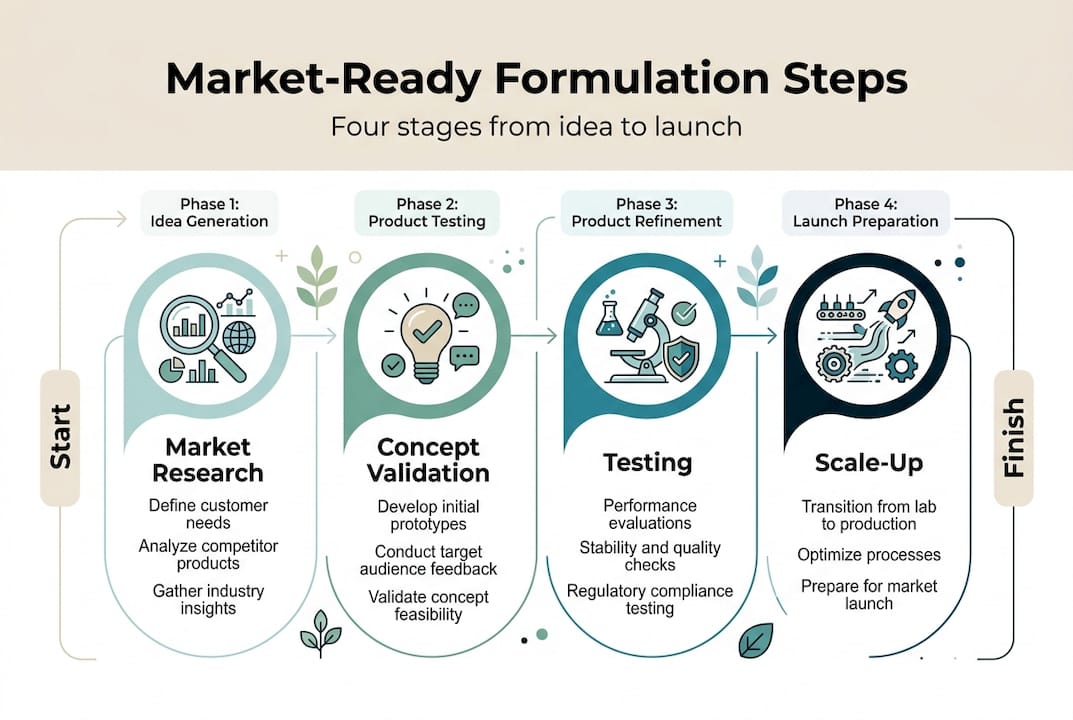
Step 1: Market analysis and idea validation
Before a single ingredient hits a beaker, you need to know whether your concept has a real audience. This step is where most failed projects cut corners. Market and consumer analysis paired with competitive benchmarking are the most overlooked first steps in product development, and skipping them is one of the clearest predictors of launch failure.
Start by mapping the competitive landscape. You want to know what's already on shelves, what claims competitors are making, and where the preference gaps are. A preference gap is the space between what consumers say they want and what current products actually deliver. That gap is your opportunity.
Here are the core tools and sources to build your market picture:
- Retail sales data (SPINS, Nielsen, or IRI for food and wellness)
- Consumer sentiment platforms (Mintel, Euromonitor, or social listening tools)
- Competitor ingredient analysis (label scraping, patent databases)
- Direct consumer feedback (surveys, focus groups, beta panels)
- Trend forecasting reports (WGSN, Trendalytics for beauty)
Once you have that data, run a simple benchmarking comparison to see where your concept stacks up:
| Evaluation factor | Your concept | Competitor A | Competitor B |
|---|---|---|---|
| Target consumer segment | Define clearly | Broad market | Niche wellness |
| Key functional claim | Unique angle? | Standard claim | Premium claim |
| Price positioning | Competitive? | Mid-range | Premium |
| Regulatory risk level | Low/medium/high | Low | Medium |
With this table filled in, you can spot where your concept is differentiated and where it might struggle. Concept validation doesn't need to be expensive. A structured online survey with 200 to 400 respondents in your target demographic, combined with two or three moderated focus groups, gives you enough signal to decide whether to move forward. Use the formulation development process as your backbone from this point forward, so every decision ties back to validated consumer need.
Step 2: Formulation design and prototyping
With a validated concept in hand, it's time to translate market insight into actual formulas. This is where science meets strategy, and the approach you choose here will directly affect your timeline, cost, and success rate.

Traditional formulation relies on iterative bench work, trial and error, and expert intuition. It works, but it's slow and expensive. Data-driven formulation using AI and machine learning tools can reduce your prototype count by 70 to 90% and cut development cycles by 50 to 60%. That's not a marginal improvement. That's the difference between a 12-month runway and a 4-month one.
| Approach | Speed | Cost | Accuracy | Best for |
|---|---|---|---|---|
| Traditional bench formulation | Slow | High | Moderate | Novel textures, artisan products |
| AI/ML-assisted formulation | Fast | Lower | High | Scalable CPG, supplements, skincare |
| Hybrid method | Moderate | Moderate | High | Complex functional products |
One important constraint to keep in mind: natural and clean label trends significantly limit your ingredient options, even as consumer demand for them grows. Your formulation model needs to account for this tension early, not after you've built a prototype around a synthetic stabilizer your target customer won't accept.
Here's a practical sequence for moving from concept to first batch:
- Define your functional targets (texture, efficacy, shelf life, flavor profile)
- Select candidate ingredients based on market data and regulatory status
- Run computational or AI-assisted screening to narrow combinations
- Build bench-top prototypes at 100 to 500g scale
- Run internal sensory evaluation against your benchmarking criteria
- Refine and iterate based on sensory scores and stability data
- Finalize formula before moving to pilot batch
For brands working on prototyping for supplements, this sequence is especially important because bioavailability and ingredient interaction data need to be baked in from the start.
Pro Tip: Always start at bench-top scale before committing to pilot or industrial production. Scaling a flawed formula costs 10 to 20 times more to fix than catching the issue at the bench.
Step 3: Safety, sensory, and regulatory testing
A great-tasting, well-formulated product that fails a safety review doesn't launch. Testing is not a formality. It's the phase that separates products that make it to shelf from those that get recalled or blocked at the border.
Every formula needs to pass a core battery of tests before you can move toward production. Here's what that looks like across categories:
- Safety assessment: Toxicological review of each ingredient and the finished formula
- Microbiological testing: Challenge testing for pathogens, yeast, and mold
- Stability testing: Accelerated and real-time studies for shelf life validation
- Sensory evaluation: Trained panel or consumer panel scoring for taste, texture, appearance, and aroma
- Nutritional analysis (food and supplements): Verified against label claims
- Compatibility testing: Ingredient interactions and packaging compatibility
For cosmetics specifically, FDA guidelines require that products must not be adulterated or misbranded, and safety must be substantiated through toxicology data or existing safety literature. This isn't optional, and it applies whether you're a startup or an established brand.
Regulatory compliance is non-negotiable. The majority of major product recalls trace back to documentation failures or skipped safety steps that seemed minor at the time.
Understanding your regulatory considerations by category and market is critical before you finalize any formula. Rules differ between FDA-regulated cosmetics, dietary supplements under DSHEA, and food products under FSMA. Getting this wrong costs far more than getting it right the first time.
Pro Tip: Document every formulation decision, every test result, and every ingredient sourcing record as you go. Audit-readiness isn't something you build retroactively. It's a habit you build from day one.
Step 4: Scale-up, packaging, and launch monitoring
Passing testing is a milestone, not a finish line. Moving from a validated bench formula to a product that performs consistently at production scale requires a disciplined process. Skipping steps here is where brands lose money fast.
Follow this sequence for a controlled scale-up:
- Pilot batch production (10 to 100x bench scale): Verify process parameters hold
- In-process quality checks: Monitor critical control points throughout the run
- Packaging validation: Confirm compatibility between formula and container
- Regulatory label review: Verify all claims, ingredient listings, and required disclosures
- Final stability confirmation: Run real-time stability on packaged product
- Production run: Full-scale manufacturing with documented batch records
Packaging is not just aesthetic. Material compatibility, barrier properties, and sustainability credentials all affect both product integrity and consumer perception. An industrialization and post-launch monitoring strategy that treats packaging as an afterthought often results in costly reformulations after launch.
Once your product is live, the work continues. Use this framework to stay on top of performance:
| Post-launch action | Frequency | Why it matters |
|---|---|---|
| Consumer feedback review | Weekly (first 90 days) | Catch sensory or efficacy issues early |
| Retail sell-through tracking | Bi-weekly | Identify distribution or pricing problems |
| Competitor monitoring | Monthly | Stay ahead of market shifts |
| Regulatory update review | Quarterly | Avoid compliance gaps as rules evolve |
| Formula stability check | Per stability protocol | Confirm shelf life claims hold |
Use your scale-up checklist to make sure nothing falls through the cracks between pilot and full production. Continuous improvement after launch isn't just best practice. It's how brands build lasting market position.
Our perspective: Why a data-driven approach beats guesswork
Here's something most formulation guides won't tell you: the most expensive mistakes in product development don't happen during manufacturing. They happen in the first two weeks of a project, when teams skip the data and go straight to the bench based on gut feel or trend reports alone.
We've seen brands spend six figures on a prototype that a proper preference gap analysis would have killed in week one. The formula was technically sound. The market just didn't want it.
Data-driven approaches consistently deliver 50 to 60% faster development timelines and 30 to 60% lower costs compared to purely empirical methods. But the bigger advantage isn't speed or cost. It's the reduction in late-stage failure. When you benchmark sensory scores and compliance criteria before you finalize a formula, you're not just saving time. You're eliminating entire categories of risk.
The brands winning in beauty, wellness, and food right now are the ones treating AI-aided formulation and regulatory intelligence as core competencies, not outsourced afterthoughts. Build those capabilities into your process from the start, and you'll spend less time fixing problems and more time scaling what works. That's the real competitive edge, and it's available to any brand willing to follow the data.
Explore formulation process insights to see how structured, intelligence-led development changes outcomes at every phase.
Streamline your formulation journey with FormlyPro
Putting this entire process into practice is significantly easier when you have the right infrastructure behind you. The FormlyPro platform is built specifically for brand owners and product developers who want to move faster without cutting corners.
FormlyPro covers every phase covered in this guide: market and competitor analysis, AI-assisted prototyping, compliance tracking across FDA and international standards, packaging design with an integrated AI mockup tool, and post-launch performance monitoring. The 8-phase product development plan walks you from raw concept through production with full analytics at every step. If you're serious about building formulations that are compliant, market-ready, and built to last, FormlyPro gives you the tools to do it without guesswork.
Frequently asked questions
What tools are essential for formulating new products?
You'll need ingredient databases, market and consumer analysis tools, prototyping equipment, sensory testing protocols, and compliance checklists tailored to your product category and target market.
How does AI or data-driven design improve formulation success?
AI and machine learning tools reduce prototypes by 70 to 90%, cut development cycles by 50 to 60%, and lower costs by 30 to 60% while improving accuracy and market fit compared to traditional methods.
What are the main regulatory requirements for cosmetics and supplements?
Under FDA guidelines, products must not be adulterated or misbranded, safety must be substantiated through toxicology or existing data, and labeling must meet all required disclosure standards for the category.
What post-launch monitoring is needed?
Monitor consumer feedback weekly in the first 90 days, track retail performance bi-weekly, review post-launch performance data monthly, and check for regulatory updates quarterly to keep your product compliant as rules evolve.
