TL;DR:
- Formulation compliance is essential and involves continuous monitoring of ingredients, labeling, and safety standards.
- Building a detailed ingredient checklist early reduces costly reformulations and last-minute regulatory issues.
- Viewing compliance as an innovation enabler helps brands develop differentiated products within regulatory boundaries.
A product is weeks from launch. The packaging is finalized, the marketing campaign is live, and then a regulatory review flags three prohibited ingredients in the formula. The launch stalls. The reformulation costs mount. The window for a seasonal release closes. This scenario plays out more often than most brand managers want to admit, and it is almost always preventable. Formulation compliance is not a box to check at the end of development. It is the backbone of every successful cosmetics and personal care product launch. This guide walks you through exactly what compliance requires, how to build a practical checklist, and how to execute a launch without costly surprises.
Table of Contents
- Understanding formulation compliance requirements
- Preparation: Building an ingredient compliance checklist
- Step-by-step process: Ensuring compliant formulation
- Common mistakes and troubleshooting compliance
- Why compliance should guide innovation, not hinder it
- Streamline your compliance process with FormlyPro
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Compliance prevents delays | Rigorous formulation compliance averts costly launch setbacks or recalls. |
| Use ingredient checklists | Begin every formulation with a checked, up-to-date ingredient list to ensure product safety and legal acceptance. |
| Document every step | Maintaining full compliance documentation makes regulatory audits and market entry smoother. |
| Stay alert to updates | Review compliance standards regularly and set reminders for team updates. |
| Technology accelerates compliance | Platforms like FormlyPro streamline checks and speed up product innovation. |
Understanding formulation compliance requirements
Formulation compliance means your product's ingredients, concentrations, labeling, and safety data meet every applicable regulatory standard in each market where you sell. It sounds straightforward, but the rules are layered, market-specific, and constantly shifting. Regulatory standards frequently change and must be monitored continuously, not just at the start of a project.
The two most influential regulatory bodies for cosmetics and personal care are the U.S. Food and Drug Administration (FDA) and the European Commission. The FDA regulates cosmetics under the Federal Food, Drug, and Cosmetic Act, while the EU operates under Regulation (EC) No 1223/2009. These frameworks differ significantly. The EU maintains a list of over 1,300 prohibited or restricted substances, while the FDA's restricted list is considerably shorter. If you plan to sell in both markets, the EU standard is typically the more demanding baseline.

One distinction that trips up many teams is the cosmetic versus drug classification. In the U.S., a product that claims to change the structure or function of the body (think anti-aging actives with strong efficacy claims) may be classified as an over-the-counter drug, triggering a completely different compliance pathway. Knowing your product's classification before formulation begins saves significant rework later.
The core compliance requirements you need to track include:
- Prohibited and restricted ingredients: Substances banned outright or permitted only below specific concentration limits
- Preservative and colorant approvals: Many colorants approved in the U.S. are banned in the EU and vice versa
- Labeling requirements: INCI nomenclature, country-of-origin rules, allergen declarations, and net content labeling
- Safety assessments: Required in the EU before market entry; strongly recommended everywhere
- Claim substantiation: Any efficacy claim must be supportable with data
The table below shows how key requirements compare across two major markets:
| Requirement | U.S. (FDA) | EU (EC 1223/2009) |
|---|---|---|
| Pre-market approval | Not required for cosmetics | Not required, but notification needed |
| Prohibited substances list | ~11 substances | 1,300+ substances |
| Safety assessment | Recommended | Mandatory |
| Responsible person | Manufacturer/distributor | Designated EU responsible person |
| CPNP notification | Not applicable | Required before market entry |
Using a structured compliance checklist from day one, combined with a practical compliance guide tailored to your target markets, keeps your team aligned and reduces the risk of late-stage surprises.
Preparation: Building an ingredient compliance checklist
An ingredient checklist is your first line of defense. It prevents non-compliance surprises that derail timelines and inflate costs. Building one before formulation starts, not after, is the difference between a smooth launch and a costly reformulation cycle.

Start by pulling from authoritative sources. The EU's CosIng database, the FDA's Voluntary Cosmetic Registration Program data, and the International Nomenclature of Cosmetic Ingredients (INCI) dictionary are the primary references. For restricted substances with concentration limits, always cross-reference the most current version of the relevant regulation, since limits are updated periodically.
Here is a comparison of the most widely used ingredient screening tools:
| Tool | Coverage | Best for | Cost |
|---|---|---|---|
| EU CosIng | EU regulations | EU market compliance | Free |
| FDA Cosmetic Database | U.S. regulations | U.S. market screening | Free |
| EWG Skin Deep | Safety ratings | Consumer-facing risk review | Free/paid tiers |
| FormlyPro | Multi-market compliance | Full workflow integration | Subscription |
| CosDNA | Ingredient function/safety | Quick ingredient lookups | Free |
Once you have your sources, build your checklist using this sequence:
- List every raw ingredient in your formula by INCI name, including carriers and preservatives
- Cross-reference each ingredient against the prohibited and restricted lists for every target market
- Document concentration limits and confirm your formula stays within them
- Flag any dual-use ingredients that may trigger drug classification in certain markets
- Record the regulatory reference for each decision so your audit trail is complete
- Assign a review owner for each ingredient category to ensure accountability
Pro Tip: Set a calendar reminder to re-screen your ingredient list every quarter. Regulatory bodies update restricted substance lists without fanfare, and a formula that was compliant six months ago may not be today.
For teams working toward a full market launch, reviewing market-ready formulation steps alongside your checklist helps you align compliance work with the broader development timeline.
Step-by-step process: Ensuring compliant formulation
With your checklist in hand, the real work begins. Compliance is not a single review at the end of formulation. It is a continuous process woven into every phase of development.
Follow this workflow to keep compliance on track from first prototype to final batch:
- Pre-formulation screen: Before your formulator selects any ingredient, run it through your checklist. Eliminate non-starters early.
- Prototype documentation: Log every ingredient, concentration, supplier, and lot number for each prototype batch. This becomes your compliance record.
- In-process review: At each formulation iteration, re-check any changed or substituted ingredient against your restricted lists.
- Lab validation: Conduct stability testing, challenge testing (for preservative efficacy), and any required safety assessments. Document results formally.
- Label review: Before finalizing packaging, verify that every label element meets the requirements of each target market, including allergen declarations and INCI listings.
- Pre-launch audit: Run a final compliance audit against your checklist, sign off with a responsible person, and archive all records.
Documenting each step with compliance checks is vital because regulators and retail partners increasingly require audit-ready records before they will list a product.
Critical note: Never rely solely on a supplier's safety data sheet as proof of compliance. Verify ingredient status independently against the current regulatory text for every target market before launch.
Pro Tip: Digital platforms that integrate formulation analytics create automatic audit trails, flag compliance issues in real time, and make it far easier to respond to retailer or regulatory requests without scrambling through spreadsheets.
Traceability matters more than most teams realize until they face a recall or a retailer audit. Building it in from the start costs almost nothing compared to reconstructing records under pressure.
Common mistakes and troubleshooting compliance
Even disciplined teams run into compliance problems. The good news is that most failures come from a small set of recurring mistakes, all of which are fixable once you know what to look for.
Outdated ingredient lists
Missed updates in regulatory standards frequently trigger costly recalls, and the root cause is almost always an ingredient list that was not re-checked after a regulatory revision.
- Fix: Assign a compliance owner who monitors regulatory bulletins from the FDA, European Commission, and any other relevant authority on a monthly basis
- Fix: Use a platform that flags regulatory changes automatically rather than relying on manual checks
Missing or incomplete records
Incomplete documentation is the second most common failure point, especially when teams change or suppliers are swapped mid-development.
- Fix: Standardize a documentation template at the start of every project and make it mandatory for every formulation change
- Fix: Store records in a centralized, version-controlled system so nothing gets lost in email threads
Ambiguous or non-compliant labeling
Labeling errors are surprisingly common, particularly for brands launching in multiple markets simultaneously. A claim that is fine in the U.S. may be regulated as a drug claim in the EU.
- Fix: Build a label review step into your compliance checklist and have it reviewed by someone familiar with each target market's rules
- Fix: Cross-check all efficacy claims against the substantiation data you have on file
Pro Tip: Schedule a formal compliance review every time you enter a new market, change a supplier, or modify a formula. These three triggers account for the majority of post-launch compliance failures.
If you do discover a compliance issue after launch, act quickly. Notify your regulatory contact, assess whether a voluntary recall or relabeling is needed, and document every corrective action. Understanding why compliance matters at a business level helps leadership make faster decisions when problems surface.
Why compliance should guide innovation, not hinder it
Here is a perspective that most compliance conversations miss: the brands that treat regulatory requirements as a creative constraint, rather than a bureaucratic obstacle, consistently out-innovate their competitors.
When your formulation team knows the boundaries early, they stop wasting cycles on ingredients that will never make it to market. They start exploring what is possible within the rules, and that is where genuinely differentiated products get built. Compliance-first R&D is not slower. It is actually faster because you eliminate dead ends before they consume resources.
There is also a competitive intelligence angle that most brands overlook. Regulatory databases and ingredient restriction lists are public. If you read them the way a strategist reads a market map, you can spot ingredient trends before they become mainstream, identify gaps your competitors have not filled, and position your formulations ahead of upcoming restrictions. Using formulation analytics to accelerate R&D turns compliance data into a genuine innovation signal.
The brands winning in 2026 are not the ones avoiding compliance. They are the ones who have made it a core part of how they think about product development from day one.
Streamline your compliance process with FormlyPro
Formulation compliance does not have to mean endless spreadsheets and last-minute regulatory scrambles. FormlyPro is built specifically for cosmetics and personal care brands that need to move fast without cutting corners on compliance.
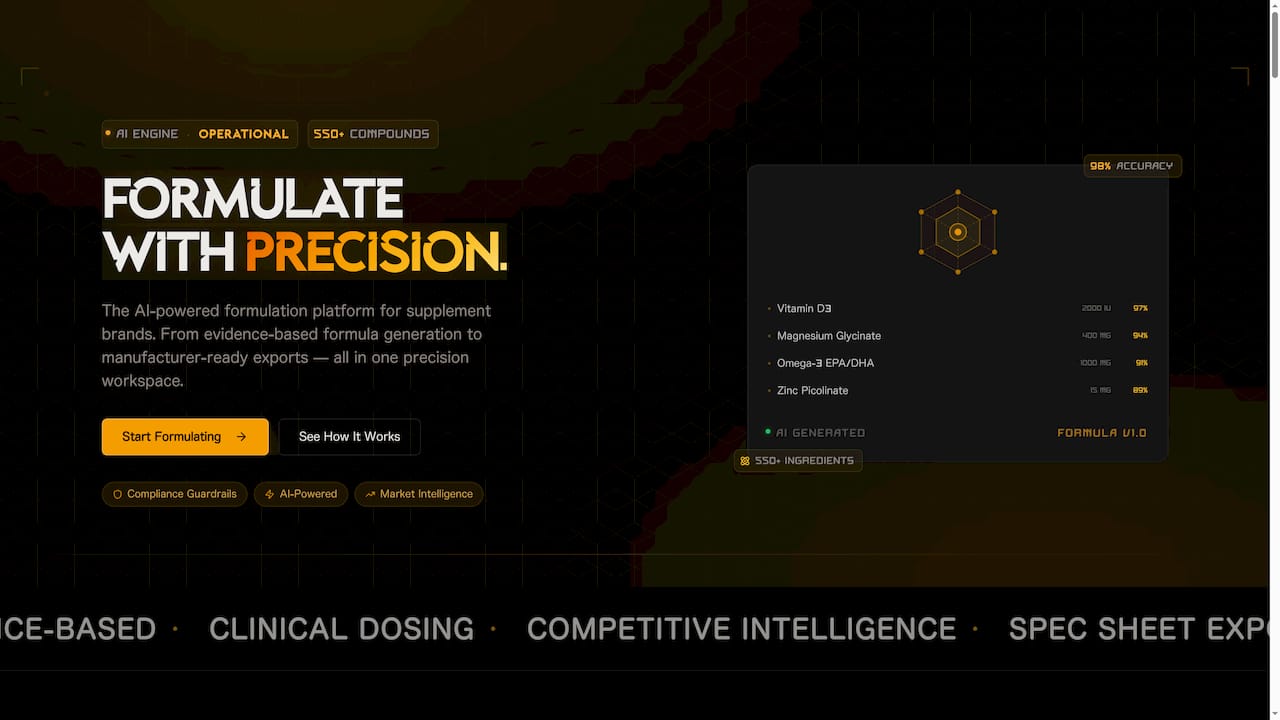
The platform guides you through an 8-phase product development process, from ideation through production, with built-in compliance checks at every stage. You get competitor formulation analysis, market research, ingredient screening, and an AI-powered packaging mockup designer, all in one place. Whether you are launching your first product or scaling a portfolio, FormlyPro gives your team the structure and data to launch confidently and compliantly.
Frequently asked questions
What are the main regulatory bodies for cosmetic formulation compliance?
Key authorities include the FDA (United States) and the European Commission (EU), which set ingredient, labeling, and safety requirements for cosmetics. Regulatory standards require ongoing monitoring because both bodies update their requirements regularly.
How often should ingredient compliance checklists be updated?
Checklists should be reviewed at least quarterly and immediately after any regulatory change or formula modification. Ingredient lists are often the source of compliance failures when teams skip re-verification after updates.
What tools help verify cosmetic ingredient compliance?
Ingredient databases like EU CosIng, compliance platforms like FormlyPro, lab testing, and official regulatory guidance documents are the most reliable options. Technology platforms can speed compliance analysis by 50%, making them a practical investment for growing brands.
Can compliance errors be reversed after product launch?
Yes, but recovery typically requires a product recall, relabeling, or full reformulation, all of which are expensive and damaging to brand trust. Missed regulatory updates frequently trigger costly recalls, which is why prevention is always the better strategy.
