TL;DR:
- Over 74% of recalls are caused by labeling errors and undeclared allergens.
- Compliance ensures products meet legal standards and builds consumer trust.
- Using proper systems and tools streamlines compliance from formulation to packaging.
Labeling errors and undeclared allergens account for over 74% of recalls in the consumer packaged goods space, and a single recall can cost a brand millions in lost inventory, legal fees, and shattered consumer trust. For brand owners launching new products, compliance is not a bureaucratic afterthought. It is the foundation that determines whether your product reaches store shelves, stays there, and builds a loyal customer base. This guide walks you through what industry compliance actually means, what the rules demand, how US and EU standards compare, and which practical strategies will keep your brand protected and positioned for growth.
Table of Contents
- What does industry compliance mean for consumer goods?
- Essential labeling and packaging compliance requirements
- US vs. EU compliance: Key similarities and differences
- Practical strategies and tools for meeting compliance standards
- Why proactive compliance is your most valuable brand asset
- Power your compliance strategy with the right tools
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Compliance drives trust | Meeting industry standards protects your reputation and wins customer loyalty. |
| Labeling is high risk | Most recalls stem from labeling errors or undeclared allergens, making accuracy essential. |
| Global rules differ | US and EU compliance approaches diverge—brands must adapt for each market. |
| Tech makes compliance easier | Digital platforms and AI tools help businesses centralize, check, and document compliance efficiently. |
What does industry compliance mean for consumer goods?
Industry compliance refers to the full set of legal requirements and voluntary standards your product must meet before it can be manufactured, sold, or marketed. For consumer goods brands, this spans two broad categories: mandatory regulations enforced by government agencies, and voluntary industry standards set by trade associations or certification bodies.
In the US, the primary regulatory bodies are the FDA, FTC, and USDA. Each governs different product categories and aspects of your business. The FDA oversees food, dietary supplements, cosmetics, and over-the-counter drugs. The FTC regulates marketing claims and advertising accuracy. The USDA handles organic certification and meat or dairy products. Key CPG requirements include accurate ingredient lists, allergen declarations, Nutrition Facts panel accuracy, and adherence to FALCPA, the Food Allergen Labeling and Consumer Protection Act.
For cosmetics brands specifically, the landscape shifted significantly with MoCRA requirements, the Modernization of Cosmetics Regulation Act of 2022, which added facility registration, product listing, and adverse event reporting to the compliance checklist.
Non-compliance carries real consequences. Regulatory agencies can issue warning letters, mandate product recalls, impose fines, or block import and export of goods. Beyond the financial hit, a public recall signals to consumers that your brand cannot be trusted, and that reputational damage can outlast any fine.
Here is a quick breakdown of the types of compliance required by product category:
- Food and beverages: Nutrition Facts, allergen labeling, FALCPA, ingredient accuracy, net weight
- Dietary supplements: Structure/function claims, Supplement Facts panel, cGMP manufacturing standards
- Cosmetics: Ingredient disclosure, warning statements, MoCRA facility registration
- Household products: Safety warnings, CPSC standards, hazardous material labeling
- Organic or USDA-certified products: Certified ingredient sourcing, approved substance lists
Compliance is not just about avoiding penalties. It is the clearest signal you can send to retailers, distributors, and consumers that your product is safe, honest, and ready for market. Brands that treat it as a core function, not a legal checkbox, consistently outperform those that do not. For a deeper look at why this matters, see compliance for food and supplement brands.
Essential labeling and packaging compliance requirements
Once you understand who regulates your product, the next step is knowing exactly what your label and packaging must include. Getting this right from the start saves you from expensive redesigns and delayed launches.
Every US consumer product label has two key panels. The Principal Display Panel (PDP) is the front-facing surface that must include the product identity and net quantity of contents. The Information Panel sits adjacent to the PDP and carries the ingredient list, allergen declarations, manufacturer information, and any required warnings.

For food products, FALCPA requires that the nine major allergens (milk, eggs, fish, shellfish, tree nuts, peanuts, wheat, soybeans, and sesame) be clearly declared, either within the ingredient list or in a separate "Contains" statement. For cosmetics, ingredient lists must follow INCI naming conventions, and any risk-related warnings such as flammability must appear prominently.
Packaging materials themselves are also regulated. FDA food-contact regulations require that packaging materials not transfer harmful substances to food, with specific migration limits in place. This applies regardless of whether the material is marketed as "natural" or "plant-based." A compostable pouch still needs to meet FDA food-contact standards before it touches your product.
Beyond federal rules, state-level packaging rules like California's Prop 65 and Extended Producer Responsibility (EPR) laws add another layer. EPR programs require brands to take financial responsibility for the end-of-life management of their packaging, and several states have active or pending legislation in 2026.
Pro Tip: Never assume a sustainable or "clean" packaging material is automatically compliant. Always request a Letter of Guarantee (LOG) from your supplier confirming FDA compliance for any food-contact surface.
Here is a summary table of core label requirements by product type:
| Product type | Key label requirements |
|---|---|
| Food and beverage | Nutrition Facts, allergen statement, ingredient list, net weight |
| Dietary supplement | Supplement Facts, serving size, structure/function claim disclaimer |
| Cosmetic | INCI ingredient list, net quantity, warnings, MoCRA registration |
| Household cleaner | Hazard warnings, first aid, CPSC compliance |
| Organic product | USDA seal, certified ingredient sourcing disclosure |
Steps for verifying packaging compliance for a new product:
- Identify your product category and the applicable federal agency
- Map all required label elements for that category
- Collect supplier LOGs or Certificates of Analysis for all packaging materials
- Cross-reference your label draft against a packaging compliance checklist
- Review state-specific rules for each market you plan to enter
- Run a final label audit before production approval
US vs. EU compliance: Key similarities and differences
If you plan to sell internationally, understanding how regulatory frameworks differ between the US and EU is critical. Assuming one market's approval transfers to another is one of the most expensive mistakes a growing brand can make.
The most fundamental difference is the regulatory philosophy. The US operates on a risk-based system using GRAS (Generally Recognized as Safe) designations and post-market enforcement. This means ingredients can be used unless proven harmful. The EU takes a precautionary approach, requiring pre-market authorization and using positive lists of approved substances. If an ingredient is not on the EU's approved list, it cannot be used, full stop.

For cosmetics, EU Cosmetics Regulation 1223/2009 requires a designated Responsible Person in the EU, a full product safety assessment, rigorous labeling in local languages, and notification through the CPNP portal before a product can be sold. The US has no equivalent pre-market notification requirement for cosmetics under traditional FDA rules, though MoCRA is narrowing that gap.
Allergen labeling also diverges significantly. The US mandates labeling for 9 major allergens, while the EU requires disclosure for 14. Celery, lupin, molluscs, mustard, and sulphur dioxide are required declarations in the EU but not in the US. This means a label fully compliant in the US may still be illegal in Europe.
Here is a comparison of key features across both markets:
| Feature | US standard | EU standard |
|---|---|---|
| Approval model | GRAS, post-market enforcement | Pre-market authorization, positive lists |
| Major allergens required | 9 | 14 |
| Cosmetic pre-notification | Not required (MoCRA adds registration) | Required via CPNP |
| Additive regulation | Risk-based assessment | Approved substance lists only |
| Food contact materials | Migration limits, FDA approval | Regulation (EC) No 1935/2004 |
Common pitfalls when switching between US and EU markets:
- Using US-approved additives that are banned in the EU
- Forgetting to translate and localize labels for EU member states
- Missing the Responsible Person requirement for cosmetics
- Overlooking EU-specific allergen declarations
- Assuming GRAS status equals EU approval
For a practical breakdown of how to build compliance into your development process from day one, see compliance in product development. You can also explore global regulatory contrasts for a deeper academic perspective on these diverging frameworks.
Practical strategies and tools for meeting compliance standards
Compliance becomes far less overwhelming when you build systems around it rather than treating it as a last-minute review. The brands that consistently avoid recalls and launch faster are the ones that operationalize compliance from the very beginning of product development.
The first step is centralizing your data. Centralized PLM or PIM systems with built-in approval workflows give your team a single source of truth for ingredient data, supplier documentation, and label versions. When everyone works from the same verified data, the risk of errors slipping through drops dramatically.
AI-powered tools are now capable of scanning label copy for allergen omissions, claim accuracy, and formatting errors in seconds. These tools do not replace a regulatory expert, but they catch the obvious mistakes before they reach production. For a broader look at how formulation analytics benefits your innovation pipeline, the efficiency gains extend well beyond compliance.
For entrepreneurs and smaller brands, the FDA provides guidance on small business exemptions for certain cosmetic requirements, but these exemptions do not eliminate your safety responsibilities. Supplier guarantees and regular audits are non-negotiable regardless of your company size.
Pro Tip: Schedule a mock recall drill at least once a year. Trace a hypothetical batch from production through distribution and identify every gap in your documentation. Most brands discover critical traceability weaknesses only after a real recall forces the issue.
Steps to create a compliance-first product launch process:
- Assign a compliance owner at the start of every new product project
- Build regulatory review into your formulation approval stage, not after
- Use AI for label compliance checks before artwork is finalized
- Require supplier LOGs for every raw material and packaging component
- Conduct a pre-launch audit against your target market's specific requirements
- Document everything, including version history for formulas and labels
For a step-by-step walkthrough of building a compliant product from scratch, the market-ready formulation steps guide covers the full process in detail.
Why proactive compliance is your most valuable brand asset
Most brands treat compliance as a cost center. They allocate budget to it reluctantly, address it reactively, and measure success by avoiding fines. That mindset leaves enormous value on the table.
The brands winning in competitive CPG categories are using compliance as a trust signal. When your label is accurate, your allergen declarations are clear, and your packaging materials are certified, you are communicating something powerful to retailers and consumers: we take this seriously. That is a competitive advantage that no marketing campaign can manufacture.
Proactive compliance through AI and automation does more than reduce errors. It accelerates your speed to market by removing last-minute regulatory bottlenecks. Brands that adopt the strictest global standards, including EU pre-market requirements, often find that US compliance becomes easier by default, not harder.
The uncomfortable truth is that most compliance failures are not caused by ignorance of the rules. They are caused by poor systems, rushed timelines, and siloed teams. Fixing those organizational issues is what separates brands that scale from brands that stall. Use your compliance checklist not as a final gate, but as a living document embedded in every phase of your product development cycle.
Power your compliance strategy with the right tools
Managing compliance across formulations, packaging, and multiple markets is genuinely complex. But the right technology makes it systematic, traceable, and far less stressful.
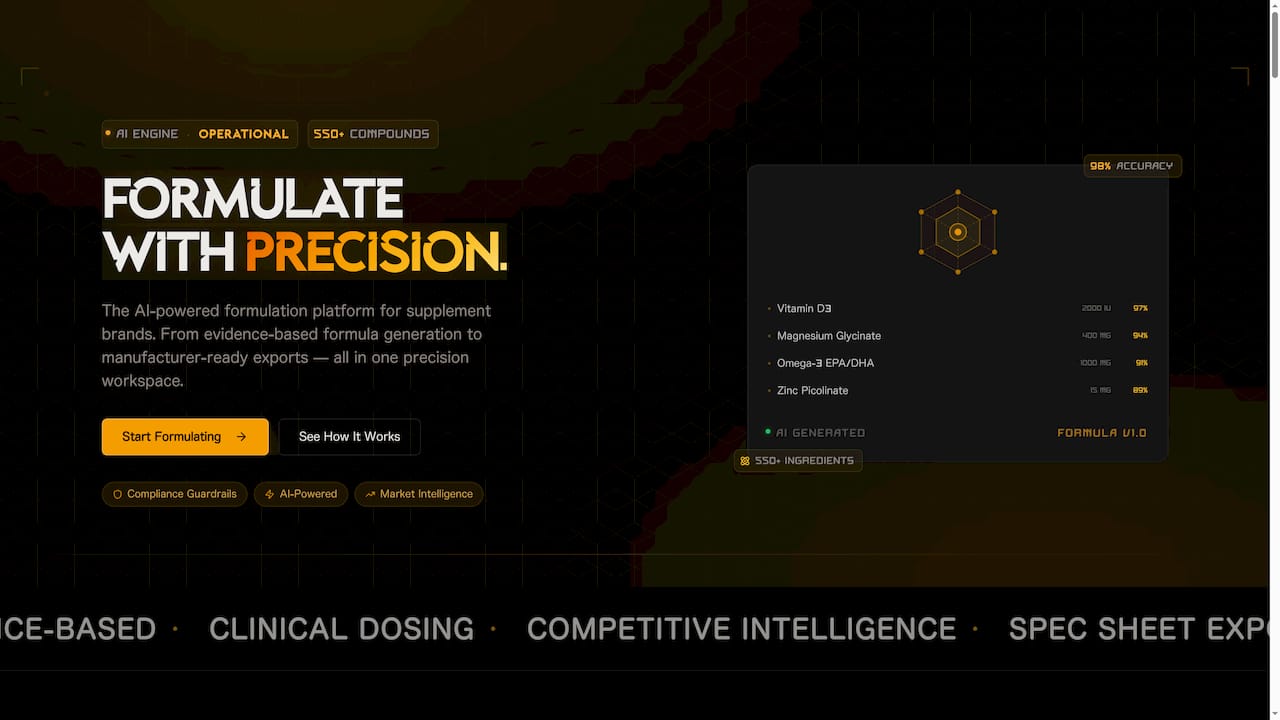
FormlyPro is built specifically for brand owners navigating this complexity. The platform guides you through an 8-phase product development process that integrates compliance at every stage, from formulation and prototyping through to packaging and production. With built-in competitor analysis, market research, and an AI-powered packaging mockup designer, you get everything you need to launch a compliant, market-ready product without juggling a dozen disconnected tools. Explore how FormlyPro can streamline your compliance workflow and help you launch with confidence.
Frequently asked questions
What are the most common compliance errors for consumer goods brands?
Undeclared allergens and labeling errors are the primary drivers of product recalls, accounting for the majority of enforcement actions across food, supplement, and cosmetic categories. Catching these errors before production requires systematic label audits and centralized ingredient data.
What documentation do I need to prove my packaging is compliant?
You typically need supplier Letters of Guarantee confirming FDA compliance for all food-contact materials, along with Certificates of Analysis and detailed records for every packaging component. These documents are your primary defense in the event of an audit or recall.
How do US and EU allergen labeling standards differ?
The US requires labeling for 9 major allergens while the EU mandates disclosure for 14, including celery, lupin, and mustard that are not required in the US. A product fully compliant in the US may still require label changes before it can be sold in European markets.
How can smaller brands keep up with evolving compliance laws?
Smaller brands should start by centralizing supplier data, securing LOGs from every supplier, and running regular internal audits to catch gaps before regulators do. Digital platforms designed for CPG compliance can make this process manageable even with a lean team.
